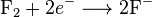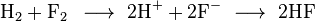Carlsberg spúšťa Web-TV bolo na mieste asi v televízny program spojiť s futbalom a humorista ostatné agendy, ktoré urobím z vás rozesmA a bezradné tiež musel Partofthegame.tv, že predložené pořad program futbalu, ktorý bol od klasických, že to bol moderný
Study group scheme flourishingThe UEFA Study Group Scheme, which heralds a new era in football technical co-operation across Europe Študijná skupina režime flourishingThe UEFA Study Group schémy, ktoré hlasatelé novej éry vo futbale technickej spolupráce-prevádzka po celej Európe
Here in web tv could also put forward the agenda in an online manner Tu v Web TV by tiež predložiť pořad v on-line spôsobom
www.partofthegame.tv

Rabu, 26 November 2008
Carlsberg spúšťa Web-TV
Selasa, 18 November 2008
India – China has dismissed talks in India by exiled Tibetan leaders on the future of the Himalayan region, saying any moves to separate Tibet from China will fail.
"Any attempt to separate Tibet from Chinese territory will be doomed," Foreign Ministry spokesman Qin Gang told a news conference Tuesday. "The so-called Tibet government in exile is not recognized by any government in the world,"
Qin's comments come as Tibetan leaders meet this week in the first major re-evaluation of their strategy since the Dalai Lama in 1988 outlined his "middle way." That philosophy pushes for autonomy but not outright independence for Tibet.
The meeting in India comes after the Dalai Lama expressed frustration over years of fruitless talks with China.
(This version CORRECTS APNewsNow. corrects spelling to Qin Gang in 2nd graf.)
Selasa, 11 November 2008
Senin, 10 November 2008
Hydrogen peroxide
Hydrogen peroxide (H2O2) is a very pale blue liquid which appears colorless in a dilute solution, slightly more viscous than water. It is a weak acid. It has strong oxidizing properties and is therefore a powerful bleaching agent that is mostly used for bleaching paper, but has also found use as a disinfectant, as an oxidizer, as an antiseptic, and in rocketry (particularly in high concentrations as high-test peroxide or HTP) as a monopropellant, and in bipropellant systems. The oxidizing capacity of hydrogen peroxide is so strong that the chemical is considered a highly reactive oxygen species.
Hydrogen peroxide is naturally produced as a byproduct of oxygen metabolism, and virtually all organisms possess enzymes known as peroxidases, which harmlessly catalytically decompose low concentrations of hydrogen peroxide to water and oxygen (see Decomposition below).
Examples of redox reactions
A good example is the reaction between hydrogen and fluorine:
We can write this overall reaction as two half-reactions: the oxidation reaction
and the reduction reaction:
Analysing each half-reaction in isolation can often make the overall chemical process clearer. Because there is no net change in charge during a redox reaction, the number of electrons in excess in the oxidation reaction must equal the number consumed by the reduction reaction (as shown above).
Elements, even in molecular form, always have an oxidation number of zero. In the first half reaction, hydrogen is oxidized from an oxidation number of zero to an oxidation number of +1. In the second half reaction, fluorine is reduced from an oxidation number of zero to an oxidation number of −1.
When adding the reactions together the electrons cancel:
And the ions combine to form hydrogen fluoride:
Redox
Redox (shorthand for reduction-oxidation reaction) describes all chemical reactions in which atoms have their oxidation number (oxidation state) changed. This can be either a simple redox process such as the oxidation of carbon to yield carbon dioxide, or the reduction of carbon by hydrogen to yield methane (CH4), or it can be a complex process such as the oxidation of sugar in the human body through a series of very complex electron transfer processes.
The term redox comes from the two concepts of reduction and oxidation. It can be explained in simple terms:
* Oxidation describes the loss of electrons / hydrogen or gain of oxygen / increase in oxidation state by a molecule, atom or ion
* Reduction describes the gain of electrons / hydrogen or a loss of oxygen / decrease in oxidation state by a molecule, atom or ion
Though sufficient for many purposes, these descriptions are not precisely correct. Oxidation and reduction properly refer to a change in oxidation number—the actual transfer of electrons may never occur. Thus, oxidation is better defined as an increase in oxidation number, and reduction as a decrease in oxidation number. In practice, the transfer of electrons will always cause a change in oxidation number, but there are many reactions which are classed as "redox" even though no electron transfer occurs (such as those involving covalent bonds).
Non-redox reactions, which do not involve changes in formal charge, are known as metathesis reactions.
Hypoxia or oxygen
Hypoxia or oxygen depletion is a phenomenon that occurs in aquatic environments as dissolved oxygen (DO; molecular oxygen dissolved in the water) becomes reduced in concentration to a point detrimental to aquatic organisms living in the system. Dissolved oxygen is typically expressed as a percentage of the oxygen that would dissolve in the water at the prevailing temperature and salinity (both of which affect the solubility of oxygen in water; see oxygen saturation and underwater). An aquatic system lacking dissolved oxygen (0% saturation) is termed anaerobic, reducing, or anoxic; a system with low DO concentration—in the range between 1 and 30% DO saturation—is called hypoxic. Most fish cannot live below 30% DO saturation. A "healthy" aquatic environment should seldom experience DO less than 80%.